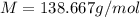An element, X, can form a chloride (XCl3) and an iodide (XI3). The chloride can be converted quantitatively into the iodide when heated and exposed to excess iodine. If 0.760 grams of XCl3 are heated with iodine, 1.610 g of XI3 are produced. What is the chemical symbol for this element? Use stoichiometry to create an equation that allows you to solve for the molecular weight of X.2 XCl3 + 3 I2 > 2 XI3 + 3 Cl2

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:10
Precision can be defined as the o exact center of a data set. o reproducibility of a measured value. o correlation between two variables that are measured in a data set agreement between a measured value and an accepted value.
Answers: 2

Chemistry, 23.06.2019 01:30
Will a solution form when the solvent and solute are both nonpolar? a. not likely b. never c. most likely
Answers: 1

Chemistry, 23.06.2019 08:00
Drag each pressure unit with the corresponding number to describe standard atmospheric pressure
Answers: 1

Chemistry, 23.06.2019 11:30
Place the following substances in order of ph from lowest ph to highest. a. neutral compounds, bases, acids b. acids, neutral compounds, bases c. bases, acids, neutral compounds d. bases, neutral compounds, acids
Answers: 1
You know the right answer?
An element, X, can form a chloride (XCl3) and an iodide (XI3). The chloride can be converted quantit...
Questions

Mathematics, 25.02.2022 04:00



Social Studies, 25.02.2022 04:10

Mathematics, 25.02.2022 04:10

Mathematics, 25.02.2022 04:10




Geography, 25.02.2022 04:10

Arts, 25.02.2022 04:10

Mathematics, 25.02.2022 04:10



Biology, 25.02.2022 04:10



Social Studies, 25.02.2022 04:20

English, 25.02.2022 04:20

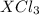 = 0.760 g
= 0.760 g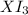 = 1.610 g
= 1.610 g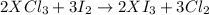
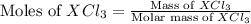
![\text{Moles of }XCl_3=\frac{0.760}{[M+3(35.5)]}](/tpl/images/0511/1010/d1e30.png)
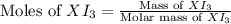
![\text{Moles of }XI_3=\frac{1.610}{[M+3(126.9)]}](/tpl/images/0511/1010/a64e9.png)
![\frac{0.760}{[M+3(35.5)]}=\frac{1.610}{[M+3(126.9)]}](/tpl/images/0511/1010/56351.png)
![\frac{0.760}{[M+106.5]}=\frac{1.610}{[M+380.7]}](/tpl/images/0511/1010/dbce0.png)