
Chemistry, 13.02.2020 22:21 viktoria1198zz
Assume that the reaction for the formation of gaseous hydrogen fluoride from hydrogen and fluorine has an equilibrium constant of 1.15 X 102 at a certain temperature. In a particular experiment, 3.00 mole of each component was added to a 1.50 L flask. Calculate the equilibrium concentrations of all species.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 02:00
Which of the following is not a good technique for managing used oil? a) have specific, labeled catch pans available for technicians who are collecting oil b) spills in your shop and any releases on pavement or outside should be poured down a drain c) do not use oil containers for antifreeze or other non-similar fluids d) be prepared for oil spills with the proper absorbents
Answers: 1

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2
You know the right answer?
Assume that the reaction for the formation of gaseous hydrogen fluoride from hydrogen and fluorine h...
Questions


English, 22.02.2021 18:50

Computers and Technology, 22.02.2021 18:50

Physics, 22.02.2021 18:50



History, 22.02.2021 18:50

History, 22.02.2021 18:50

Mathematics, 22.02.2021 18:50

English, 22.02.2021 18:50

Advanced Placement (AP), 22.02.2021 18:50


Social Studies, 22.02.2021 18:50

Arts, 22.02.2021 18:50




Arts, 22.02.2021 18:50


![[H_2]= 0.314 M](/tpl/images/0510/5228/1fffd.png)
![[F_2]=0.314 M](/tpl/images/0510/5228/5de3f.png)
![[HF]=3.372 M](/tpl/images/0510/5228/8e291.png)
![[H_2]=\frac{3.00 mol}{1.5 L}=2.0 M](/tpl/images/0510/5228/f9117.png)
![[F_2]=\frac{3.00 mol}{1.5 L}=2.0 M](/tpl/images/0510/5228/b656e.png)
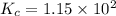
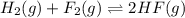
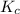 is given by :
is given by :![K_c=\frac{[HF]^2}{[H_2][F_2]}](/tpl/images/0510/5228/a2854.png)
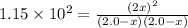
![[H_2]=(2.0 -1.686)M = 0.314 M](/tpl/images/0510/5228/64b44.png)
![[F_2]=(2.0 -1.686)M = 0.314 M](/tpl/images/0510/5228/f4a49.png)
![[HF]=(2\times 1.686)M = 3.372 M](/tpl/images/0510/5228/dc9b5.png)


