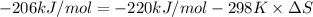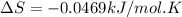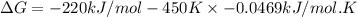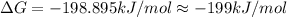For the gaseous reaction of carbon monoxide and chlorine to form phosgene (COCL2)
perform the following calculations
A) calculate delta S at 298k ( delta H= -220.kj/mol and delta G= -206kj/mol
B) assuming that delta S and delta H change little with temperature, calculate at 450k

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 07:00
Introduction of drugs into the gastrointestinal tract is a form of administration. a. enteral b. topical c. parenteral d. inhalation
Answers: 1

Chemistry, 23.06.2019 08:40
The activation energy for this reaction is 75 kj·mol–1. the enzyme catalase (found in blood) lowers the activation energy to 8.0 kj·mol–1. at what temperature would the non-catalyzed reaction need to be run to have a rate equal to that of the enzyme-catalyzed reaction at 25°c?
Answers: 2

You know the right answer?
For the gaseous reaction of carbon monoxide and chlorine to form phosgene (COCL2)
perfor...
perfor...
Questions


Computers and Technology, 02.12.2021 21:00

Mathematics, 02.12.2021 21:00


Physics, 02.12.2021 21:00

Computers and Technology, 02.12.2021 21:00





Mathematics, 02.12.2021 21:00


Mathematics, 02.12.2021 21:00

Mathematics, 02.12.2021 21:00




Mathematics, 02.12.2021 21:00

Mathematics, 02.12.2021 21:00

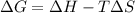
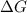 = Gibbs free energy = -206 kJ/mol
= Gibbs free energy = -206 kJ/mol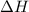 = enthalpy change = -220 kJ/mol
= enthalpy change = -220 kJ/mol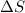 = entropy change = ?
= entropy change = ?