
Chemistry, 13.02.2020 20:02 connorgking
A. Based on the activation energies and frequency factors, rank the following reactions from fastest to slowest reaction rate, assuming they are all at the same temperature and that each starts with the same initial concentration.
E, 50 kJ/mol E,-350 kJ/mol 50 kJ/mol
A = 1.5 × 10-7 s-i A = 1.9 × 10-7 s-i A = 1.5 × 10-7 s-1
Fraction of molecules
The exponential term in the Arrhenius equation is equal to the fraction of molecules, f, with kinetic energy greater than or equal to the activation energy: f=e?Ea/(R?T). Most scientific calculators have an exfunction as the second function of the LN button.
B. A certain reaction with an activation energy of 165 kJ/mol was run at 505 K and again at 525 K . What is the ratio of f at the higher temperature to f at the lower temperature?

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
A. Based on the activation energies and frequency factors, rank the following reactions from fastest...
Questions

Advanced Placement (AP), 26.02.2021 22:10


Health, 26.02.2021 22:10



Mathematics, 26.02.2021 22:10

Mathematics, 26.02.2021 22:10

History, 26.02.2021 22:10

Mathematics, 26.02.2021 22:10



Mathematics, 26.02.2021 22:10

Mathematics, 26.02.2021 22:10



Spanish, 26.02.2021 22:10

Chemistry, 26.02.2021 22:10


Mathematics, 26.02.2021 22:10

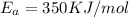 ,
, 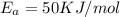 ,
, 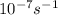 , A = 1.9×
, A = 1.9×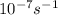 , A=1.5×
, A=1.5×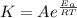
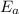 = Activation Energy
= Activation Energy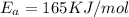
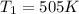
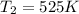
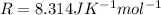
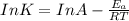
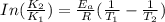
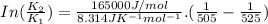
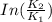 = 19846.04×7.544×
= 19846.04×7.544×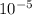 = 1.497
= 1.497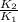 =
=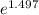 = 4.469
= 4.469 

