Chemistry, 13.02.2020 18:40 llamasking
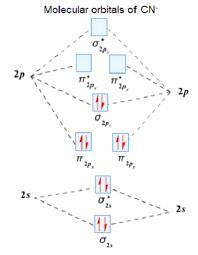
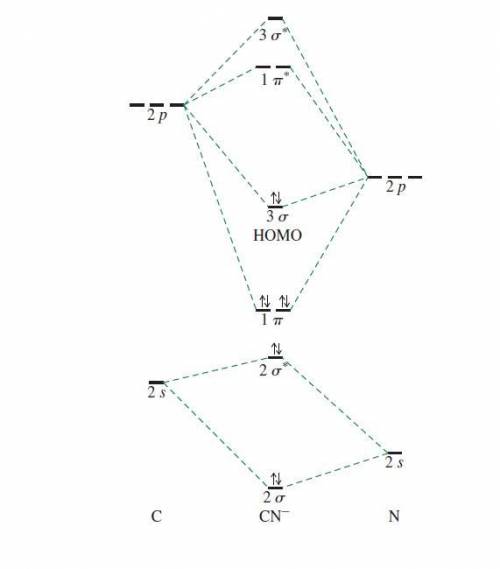
Using the molecular orbital model, write electron configurations for the following diatomic species and calculate the bond orders. Which ones are paramagnetic? Place the species in order of increasing bond length and bond energy.
a. CN +
b. CN
c. CN -

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 23.06.2019 01:50
Drag the tiles to the correct locations. each tile can be used more than once, but not all tiles will be used. one or more locations will remain empty. nitrosyl fluoride has the chemical formula nof nitrogen has five valence electrons, oxygen has six, and fluorine has seven. complete the lewis structure for this covalent compound. f n = = = . : : 0 : reset next um. all rights reserved us 2
Answers: 2

Chemistry, 23.06.2019 09:30
What is the force of an object when it landed(sitting in the ground)
Answers: 2

Chemistry, 23.06.2019 12:30
Atriple covalent bond involves two atoms sharing three pairs of electrons. true false
Answers: 2
You know the right answer?
Using the molecular orbital model, write electron configurations for the following diatomic species...
Questions

Computers and Technology, 15.06.2021 16:00


History, 15.06.2021 16:00


English, 15.06.2021 16:00

Mathematics, 15.06.2021 16:00



Mathematics, 15.06.2021 16:00



Social Studies, 15.06.2021 16:00


Chemistry, 15.06.2021 16:00

Business, 15.06.2021 16:00

Mathematics, 15.06.2021 16:00

World Languages, 15.06.2021 16:00

English, 15.06.2021 16:00