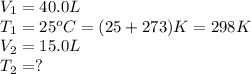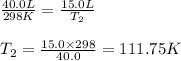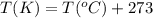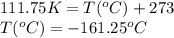Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
Ethylene, C2H4, is used to make plastic milk bottles. When the volume of 1000 g of ethylene at 25oC...
Questions

Mathematics, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

History, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

History, 16.11.2020 04:00

Arts, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00



Mathematics, 16.11.2020 04:00

Computers and Technology, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

History, 16.11.2020 04:00

English, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

Business, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

Mathematics, 16.11.2020 04:00

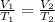
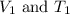 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.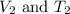 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.