
Based on enthalpy of formation data species ∆H◦ f H2S(g) −20.63 kJ/mol O2(g) 0 kJ/mol H2O(ℓ) −285.83 kJ/mol SO2(g) −296.83 kJ/mol calculate ∆Hrxn for 2 H2O(ℓ) + 2 SO2(g) ←→ 2 H2S(g) + 3 O2(g) 1. 562 kJ · mol−1 2. −562 kJ · mol−1 3. −1124 kJ · mol−1 4. 1124 kJ · mol−1

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

You know the right answer?
Based on enthalpy of formation data species ∆H◦ f H2S(g) −20.63 kJ/mol O2(g) 0 kJ/mol H2O(ℓ) −285.83...
Questions

Biology, 20.10.2021 14:00

Chemistry, 20.10.2021 14:00


Business, 20.10.2021 14:00






History, 20.10.2021 14:00




Health, 20.10.2021 14:00



English, 20.10.2021 14:00



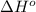
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f_{\text{(product)}}]-\sum [n\times \Delta H^o_f_{\text{(reactant)}}]](/tpl/images/0508/1357/eb0fa.png)
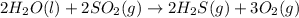
![\Delta H^o_{rxn}=[(2\times \Delta H^o_f_{(H_2S(g))})+(3\times \Delta H^o_f_{(O_2(g))})]-[(2\times \Delta H^o_f_{(H_2O(l))})+(2\times \Delta H^o_f_{(SO_2(g))})]](/tpl/images/0508/1357/e0170.png)
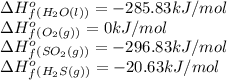
![\Delta H_{rxn}=[(2\times (-20.63))+(3\times (0))]-[(2\times (-285.83))+(2\times (-296.83))]\\\\\Delta H_{rxn}=1124kJ](/tpl/images/0508/1357/a0bfc.png)


