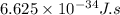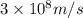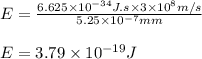Chemistry, 29.01.2020 02:44 mathman783
Electromagnetic radiation with a wavelength of 525 nm appears as green light to the human eye. the energy of one photon of this light
a) 1.04*10^-22
b)1.4*10^-31
c) 2.64*10^18
d) 3.79*10^-28
e) 3.79*10^-19

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 07:20
The diagrams show objects’ gravitational pull toward each other. which statement describes the relationship between diagram x and y? gravity attracts only larger objects toward one another. gravity attracts larger objects only if they are close to one another. if the masses of the objects increase, then the force between them also increases. if distance between the objects increases, then the amount of force also increases.
Answers: 1

Chemistry, 22.06.2019 16:00
Answer asap : ( a. how does mucus prevent the entry of pathogens? b. describe two ways white blood cells protect us from pathogens.
Answers: 1

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3
You know the right answer?
Electromagnetic radiation with a wavelength of 525 nm appears as green light to the human eye. the e...
Questions

English, 26.10.2020 17:40


English, 26.10.2020 17:40

Mathematics, 26.10.2020 17:40

History, 26.10.2020 17:40

Health, 26.10.2020 17:40

Social Studies, 26.10.2020 17:40


Mathematics, 26.10.2020 17:40

Mathematics, 26.10.2020 17:40


Mathematics, 26.10.2020 17:40



Mathematics, 26.10.2020 17:40

Biology, 26.10.2020 17:40


Geography, 26.10.2020 17:40


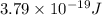
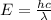
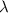 = wavelength of light =
= wavelength of light = 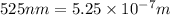 (Conversion factor:
(Conversion factor: 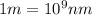 )
)