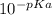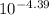Chemistry, 28.01.2020 18:51 nmartin5185
In an aqueous solution of a certain acid the acid is 4.4% dissociated and the ph is 3.03. calculate the acid dissociation constant ka of the acid. round your answer to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
What volume of a 2.00 m stock solution of naoh is needed to prepare 150. ml of 0.40 m solution?
Answers: 2

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1
You know the right answer?
In an aqueous solution of a certain acid the acid is 4.4% dissociated and the ph is 3.03. calculate...
Questions


History, 03.04.2020 00:29

Mathematics, 03.04.2020 00:29

Chemistry, 03.04.2020 00:29

Advanced Placement (AP), 03.04.2020 00:29

Mathematics, 03.04.2020 00:29

Mathematics, 03.04.2020 00:29

English, 03.04.2020 00:29

History, 03.04.2020 00:29


Mathematics, 03.04.2020 00:29



Mathematics, 03.04.2020 00:29

English, 03.04.2020 00:29




Biology, 03.04.2020 00:29

Computers and Technology, 03.04.2020 00:29