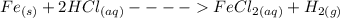Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 23.06.2019 04:00
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1

Chemistry, 23.06.2019 06:30
What is the chemical formula for a compound between li and br? libr li2br libr2 libr3
Answers: 1

You know the right answer?
Apiece of metallic iron (10 moles) was dissolved in concentrated hydrochloric acid. the reaction for...
Questions

Social Studies, 24.09.2020 02:01


History, 24.09.2020 02:01

History, 24.09.2020 02:01

Biology, 24.09.2020 02:01


Geography, 24.09.2020 02:01




English, 24.09.2020 02:01


Mathematics, 24.09.2020 02:01





English, 24.09.2020 02:01

Mathematics, 24.09.2020 02:01

Mathematics, 24.09.2020 02:01