Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1


You know the right answer?
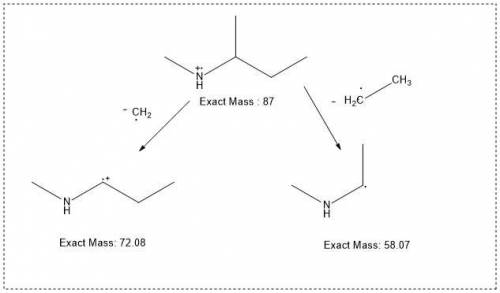
Draw a structure for an amine that exhibits a molecular ion at m+ = 87 and that produces fragments a...
Questions


Mathematics, 21.06.2019 19:30

Mathematics, 21.06.2019 19:30



Mathematics, 21.06.2019 19:30


English, 21.06.2019 19:30



Social Studies, 21.06.2019 19:30

History, 21.06.2019 19:30


Physics, 21.06.2019 19:30



Mathematics, 21.06.2019 19:30


Mathematics, 21.06.2019 19:30