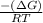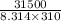Chemistry, 24.01.2020 23:31 haileyw123
Citrate synthase catalyzes the reaction: + − → + − the standard free energy change for the reaction is −31.5 ∙ −1. calculate the equilibrium constant for this reaction at 37℃.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2


Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1
You know the right answer?
Citrate synthase catalyzes the reaction: + − → + − the standard free energy change for the re...
Questions




















 citrate + HS-CoA
citrate + HS-CoA
 ) of a reaction and equilibrium constant (K) is as follows.
) of a reaction and equilibrium constant (K) is as follows.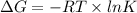
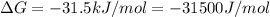 (as 1 kJ = 1000 J)
(as 1 kJ = 1000 J)