
Chemistry, 24.01.2020 04:31 bbbbriasutton
Which solution is more concentrated?
a. 2.00 moles of naoh dissolved in 1.00 liter of solution
b. 2.00 grams of naoh dissolved in 1.00 liter of solution

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2
You know the right answer?
Which solution is more concentrated?
a. 2.00 moles of naoh dissolved in 1.00 liter of s...
a. 2.00 moles of naoh dissolved in 1.00 liter of s...
Questions

Mathematics, 19.03.2021 23:50





Mathematics, 19.03.2021 23:50

Mathematics, 19.03.2021 23:50

Mathematics, 19.03.2021 23:50

Chemistry, 19.03.2021 23:50

Biology, 19.03.2021 23:50


Social Studies, 19.03.2021 23:50

Mathematics, 19.03.2021 23:50

Mathematics, 19.03.2021 23:50



Mathematics, 19.03.2021 23:50


Mathematics, 19.03.2021 23:50



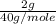 which is equal to 0.05 moles of NaOH.
which is equal to 0.05 moles of NaOH. =0.05 M
=0.05 M

