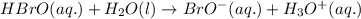Chemistry, 21.01.2020 03:31 holaadios222lol
Problem page fill in the left side of this equilibrium constant equation for the reaction of hypobromous acid with water.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1
You know the right answer?
Problem page fill in the left side of this equilibrium constant equation for the reaction of hypobro...
Questions

English, 13.04.2021 21:10


Mathematics, 13.04.2021 21:10

Mathematics, 13.04.2021 21:10

Mathematics, 13.04.2021 21:10


Mathematics, 13.04.2021 21:10




Chemistry, 13.04.2021 21:10


History, 13.04.2021 21:10


Mathematics, 13.04.2021 21:10

Mathematics, 13.04.2021 21:10


Biology, 13.04.2021 21:10


Mathematics, 13.04.2021 21:10

![K_a=\frac{[BrO^-][H_3O^+]}{[HBrO]}](/tpl/images/0463/4696/6706c.png)
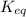
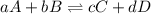
![K_{eq}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0463/4696/9c8b0.png)