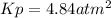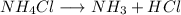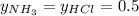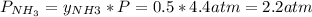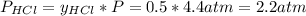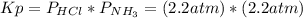Chemistry, 21.01.2020 01:31 mslamaya11
Asample of solid ammonium chloride was placed into an evacuated rigid container and then heated so that it decomposed to ammonia gas and hydrogen chloride gas. after heating the total pressure in the container was found to be 4.4 atm. calculate kp at this temperature for the decomposition reaction

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
Asample of solid ammonium chloride was placed into an evacuated rigid container and then heated so t...
Questions



Mathematics, 28.01.2021 23:20




English, 28.01.2021 23:20


Mathematics, 28.01.2021 23:20

Chemistry, 28.01.2021 23:20

Chemistry, 28.01.2021 23:20


Physics, 28.01.2021 23:20

Mathematics, 28.01.2021 23:20

Mathematics, 28.01.2021 23:20



Mathematics, 28.01.2021 23:20