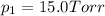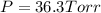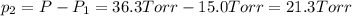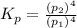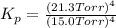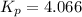Chemistry, 18.01.2020 00:31 shelbylowery789
In a study of the following reaction at 1200 k it was observed that when the equilibrium partial pressure of water vapor is 15.0 torr, the total pressure at equilibrium is 36.3 torr.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3


Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
In a study of the following reaction at 1200 k it was observed that when the equilibrium partial pre...
Questions


Mathematics, 20.05.2021 18:50

Health, 20.05.2021 18:50



English, 20.05.2021 18:50

English, 20.05.2021 18:50


Advanced Placement (AP), 20.05.2021 18:50


Social Studies, 20.05.2021 18:50

History, 20.05.2021 18:50


Chemistry, 20.05.2021 18:50

English, 20.05.2021 18:50



Mathematics, 20.05.2021 18:50


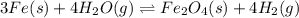
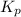 foe this reaction at 1200 K.
foe this reaction at 1200 K.