
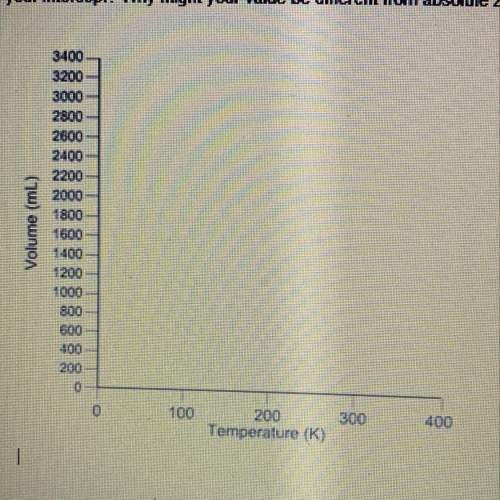
From part 2 of the lab activity, plot a graph of volume vs. temperature (in kelvins) with the two data points that resulted from your experiment. draw a straight line connecting the two points, thus assuming a linear relationship between volume and temperature. • the x-intercept corresponds to where the volume would be zero and the temperature can be no lower, defined as absolute zero. absolute zero is 0 k. compare your results to those expected. how close to absolute zero was your intercept? why might your value be different from absolute zero?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Electrons are extremely important to what area of technology? a) anti-aging research b) household product development c) electronics d) drug discovery
Answers: 3

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1
You know the right answer?
From part 2 of the lab activity, plot a graph of volume vs. temperature (in kelvins) with the two da...
Questions

Mathematics, 25.11.2020 09:30



Biology, 25.11.2020 09:30

Mathematics, 25.11.2020 09:30

Mathematics, 25.11.2020 09:30


Physics, 25.11.2020 09:30

Chemistry, 25.11.2020 09:30


Mathematics, 25.11.2020 09:30




Mathematics, 25.11.2020 09:30


Arts, 25.11.2020 09:30

Physics, 25.11.2020 09:30

Biology, 25.11.2020 09:30




