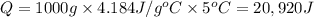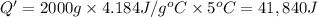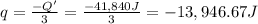Chemistry, 17.01.2020 06:31 dbn4everloved
A1.0 g sample of a cashew was burned in a calorimeter containing 1000. g of water, and the temperature of the water changed from 20.0°c to 25.0°c. in another experiment, a 3.0 g sample of a marshmallow was burned in a calorimeter containing 2000. g of water, and the temperature of the water changed from 25.0°c to 30.0°c. based on the data, which of the following can be concluded about the energy content for 1.0 g of each of the two substances? (the specific heat of water is 4.2 j/(g⋅° (a) the combustion of 1.0 g of cashew releases less energy than the combustion of 1.0 g of marshmallow. (b) the combustion of 1.0 g of cashew releases the same amount of energy as the^combustion of 1.0 g of marshmallow. (c) the combustion of 1.0 g of cashew releases more energy than the combustion of 1.0g of marshmallow. (d) no comparison can be made because the two systems started with different masses of food, different masses of water, and different initial temperatures.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2


Chemistry, 23.06.2019 05:00
Which of the following describes qualitative data? a) recording the temperature of a solid as it is warmed. b) noting the color of a solution as it is heated. c) measuring the volume of an object by water displacement. d) taking the mass of an object using a balance.
Answers: 2
You know the right answer?
A1.0 g sample of a cashew was burned in a calorimeter containing 1000. g of water, and the temperatu...
Questions


English, 20.01.2021 15:50


Medicine, 20.01.2021 16:00


English, 20.01.2021 16:00


History, 20.01.2021 16:00

English, 20.01.2021 16:00

History, 20.01.2021 16:00


Biology, 20.01.2021 16:00

Mathematics, 20.01.2021 16:00

English, 20.01.2021 16:00

English, 20.01.2021 16:00

Mathematics, 20.01.2021 16:00




Chemistry, 20.01.2021 16:00