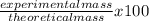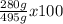Chemistry, 12.01.2020 02:31 jakobrobinette
What is the % yield when 140.0 grams of ethylene gas (c2h4) reacts with excess chlorine to form 280.0 grams of 1,2-dichloro ethane (c2h4cl2)? (given equation is c2h4 + cl2 —> c2h4cl2

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1


Chemistry, 23.06.2019 06:30
(04.01 lc) which of the following is true about science? (5 points) select one: a. it is not influenced by social conditions. b. it is not determined by external local factors. c. political conditions are unable to influence it. d. economic concerns may prevent it from solving problems.
Answers: 1
You know the right answer?
What is the % yield when 140.0 grams of ethylene gas (c2h4) reacts with excess chlorine to form 280....
Questions


Mathematics, 19.12.2019 16:31

Mathematics, 19.12.2019 16:31


Mathematics, 19.12.2019 16:31

Mathematics, 19.12.2019 16:31





Biology, 19.12.2019 16:31

Mathematics, 19.12.2019 16:31





Mathematics, 19.12.2019 17:31

English, 19.12.2019 17:31