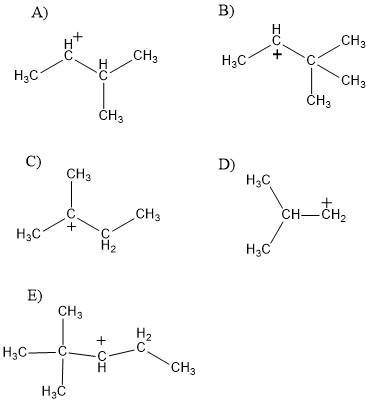
Which of the following carbocations would not be likely to undergo rearrangement?
a) c...

Chemistry, 10.01.2020 02:31 cupcake20019peehui
Which of the following carbocations would not be likely to undergo rearrangement?
a) ch3chchch3 ch3 ch3
b) ch3choch3 ch3 ch
c) ch3cch2 ch3
d) ch3 ch3chch2
e) ch3 ch3cchch2ch3 ch3

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 08:00
Determine the number of moles of air present in 1.35 l at 750 torr and 17.0°c. which equation should you use? n=pv/rt what is the number of moles present? ⇒ 0.056 mol a sample of n2 gas occupying 800.0 ml at 20.0°c is chilled on ice to 0.00°c. if the pressure also drops from 1.50 atm to 1.20 atm, what is the final volume of the gas? which equation should you use? v2= p1v1t2/p2t1 what is the final volume of the gas? ⇒ 932 ml these are the answers
Answers: 1
You know the right answer?
Questions

French, 14.12.2021 14:00

Chemistry, 14.12.2021 14:00

Social Studies, 14.12.2021 14:00


Social Studies, 14.12.2021 14:00

Social Studies, 14.12.2021 14:00

Physics, 14.12.2021 14:00

Social Studies, 14.12.2021 14:00

Social Studies, 14.12.2021 14:00

Mathematics, 14.12.2021 14:00

SAT, 14.12.2021 14:00

Chemistry, 14.12.2021 14:00



English, 14.12.2021 14:00


Mathematics, 14.12.2021 14:00

Chemistry, 14.12.2021 14:00


Arts, 14.12.2021 14:00