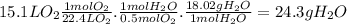What mass of water, in grams, would have to be decomposed
toproduce 15.1 l of molecular oxygen...

Chemistry, 09.01.2020 07:31 heyperdomo8496
What mass of water, in grams, would have to be decomposed
toproduce 15.1 l of molecular oxygen at stp?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

You know the right answer?
Questions

Physics, 21.06.2021 07:00

Mathematics, 21.06.2021 07:10

Chemistry, 21.06.2021 07:10

Mathematics, 21.06.2021 07:10

Mathematics, 21.06.2021 07:10



Mathematics, 21.06.2021 07:10

Mathematics, 21.06.2021 07:10

Mathematics, 21.06.2021 07:10

Mathematics, 21.06.2021 07:10


Mathematics, 21.06.2021 07:10


Mathematics, 21.06.2021 07:20





Mathematics, 21.06.2021 07:20