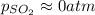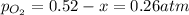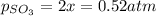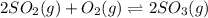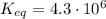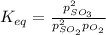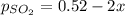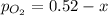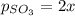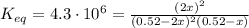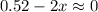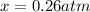Chemistry, 31.12.2019 01:31 aaliyahettorre
2so2(g) + o2(g) equilibrium reaction arrow 2 so3(g) calculate the equilibrium partial pressures of so2, o2, and so3 produced from an initial mixture in which the partial pressures of so2 and o2 = 0.52 atm and the partial pressure of so3 = 0 (exactly).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:10
Between 2014 and 2016, more than 25,000 children in flint, michigan, drank water that was contaminated with lead from lead pipes. during this time, the city claimed the water was safe to drink. which of these actions could the city have taken to ensure that the drinking water was free from lead?
Answers: 3

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2

Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1
You know the right answer?
2so2(g) + o2(g) equilibrium reaction arrow 2 so3(g) calculate the equilibrium partial pressures of s...
Questions

Mathematics, 08.12.2021 01:10



English, 08.12.2021 01:10

Biology, 08.12.2021 01:10



English, 08.12.2021 01:10


Mathematics, 08.12.2021 01:20


Mathematics, 08.12.2021 01:20

Social Studies, 08.12.2021 01:20

Mathematics, 08.12.2021 01:20

History, 08.12.2021 01:20


Mathematics, 08.12.2021 01:20

Mathematics, 08.12.2021 01:20

Mathematics, 08.12.2021 01:20

History, 08.12.2021 01:20