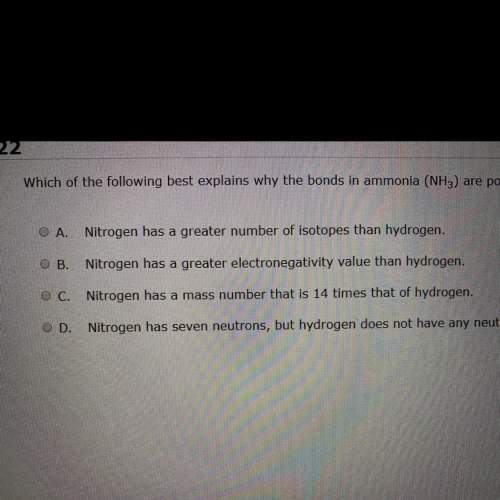
Which of the following bets explains why the bond in ammonia nh3 are polar covalent?
...

Chemistry, 29.12.2019 23:31 natalie2sheffield
Which of the following bets explains why the bond in ammonia nh3 are polar covalent?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 17:30
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

You know the right answer?
Questions





Biology, 02.08.2019 07:10


Mathematics, 02.08.2019 07:10

Mathematics, 02.08.2019 07:10

History, 02.08.2019 07:10

English, 02.08.2019 07:10

History, 02.08.2019 07:10

Social Studies, 02.08.2019 07:10

Biology, 02.08.2019 07:10

Mathematics, 02.08.2019 07:10




Mathematics, 02.08.2019 07:10

Chemistry, 02.08.2019 07:20



