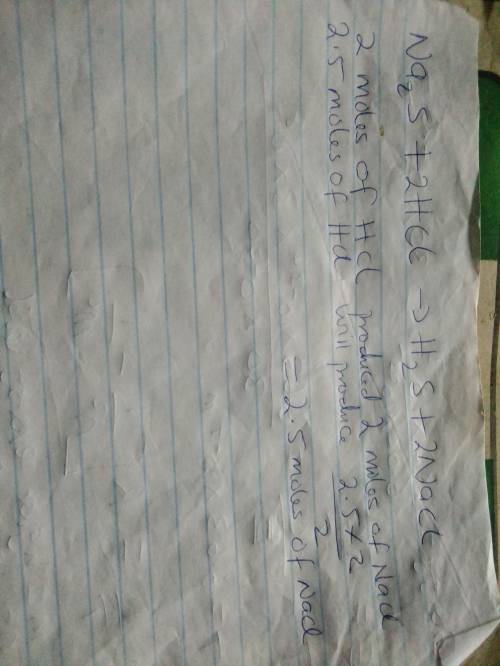Chemistry, 26.12.2019 21:31 miahsmith258
Sodium sulfide reacts with hydrochloric acid to produce hydrosulfuric acid and sodium chloride. (you need to write and balance the equation to answer the question.) how many moles of sodium chloride can be produced from 2.50 moles hydrochloric acid?

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1

Chemistry, 23.06.2019 00:30
When did stem cell research become known ? who discovered stem cell? what experiments or studies have been conducted so far?
Answers: 3

Chemistry, 23.06.2019 09:00
Agust of wind blowing east pushes against a ball. when will the wind do work on the ball? when the ball moves to the east when the ball moves to the north when the ball stays in one place when the ball moves north or south
Answers: 1

Chemistry, 23.06.2019 11:00
Find the enthalpy of neutralization of hcl and naoh. 87 cm3 of 1.6 mol dm-3 hydrochloric acid was neutralized by 87 cm3 of 1.6 mol dm-3 naoh. the temperature rose from 298 k to 317.4 k. the specific heat capacity is the same as water, 4.18 j/k g. a. -101.37 kj b. 7055 kj c. 10,1365 kj
Answers: 1
You know the right answer?
Sodium sulfide reacts with hydrochloric acid to produce hydrosulfuric acid and sodium chloride. (you...
Questions

Mathematics, 06.03.2021 04:00


Mathematics, 06.03.2021 04:00

Mathematics, 06.03.2021 04:00



Social Studies, 06.03.2021 04:00



Mathematics, 06.03.2021 04:00

History, 06.03.2021 04:00

English, 06.03.2021 04:00

Mathematics, 06.03.2021 04:00

English, 06.03.2021 04:00

Physics, 06.03.2021 04:00

Biology, 06.03.2021 04:00


Business, 06.03.2021 04:00


Mathematics, 06.03.2021 04:00