pleaeee
which of the following explains the vsepr geometry of a carbon tetrachloride mol...

pleaeee
which of the following explains the vsepr geometry of a carbon tetrachloride molecule? (5 points)
it is bent because there are four bonded pairs around carbon.
it is tetrahedral because there are four bonded pairs around carbon.
it is bent because there are two bonded pairs and two lone pairs around carbon.
it is tetrahedral because there are two bonded pairs and two lone pairs around carbon.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:00
__ _ _ _ _ is the process of removing earth materials from their original sites through weathering and transport and depositing the in another location. a. erosion b. sedimentation c. lithification d. dissolution
Answers: 1

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Questions




Arts, 01.02.2021 18:00




English, 01.02.2021 18:00



Social Studies, 01.02.2021 18:00


Mathematics, 01.02.2021 18:00


Mathematics, 01.02.2021 18:00





Computers and Technology, 01.02.2021 18:00

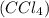 has carbon atom as central atom surrounded by four chlorine atoms.
has carbon atom as central atom surrounded by four chlorine atoms.

