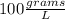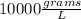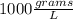Which of the following has the largest density? group of answer choices
a 1-g object w...


Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 23.06.2019 06:00
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2

Chemistry, 23.06.2019 07:30
In a laboratory determination of the atomic weight of tin, a sample of tin is weighed in a crucible. nitric acid is added, and the reaction proceeds to give a hydrated tin(iv)oxide plus no2and h2o. the hydrated tin(iv)oxide is then heated strongly and reacts as follows: sno2.xh2o(s)sno2(s)+ xh2o(g)the sno2is finally cooled and weighed in the crucible. explain the effect on the calculated atomic weight of tin that would result from each of the following experimental errors: (a)considerable spattering occurs when the nitric acid is added to the tin.(b)the hydrated tin(iv)oxide is not heated sufficiently to change it completely to tin oxide.
Answers: 2

Chemistry, 23.06.2019 08:00
Can anyone answer these questions? ? i need it before 1: 00pm today
Answers: 3
You know the right answer?
Questions


Mathematics, 14.07.2019 02:00

Physics, 14.07.2019 02:00



History, 14.07.2019 02:00






Mathematics, 14.07.2019 02:00

History, 14.07.2019 02:00

Chemistry, 14.07.2019 02:00


Mathematics, 14.07.2019 02:00

Business, 14.07.2019 02:00



Mathematics, 14.07.2019 02:00

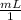 ×
× 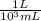
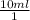 ×
×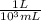 = .01L
= .01L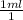 ×
×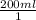 ×
×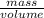 plug in the values you are given and you get
plug in the values you are given and you get