Chemistry, 19.12.2019 00:31 dgayles8761
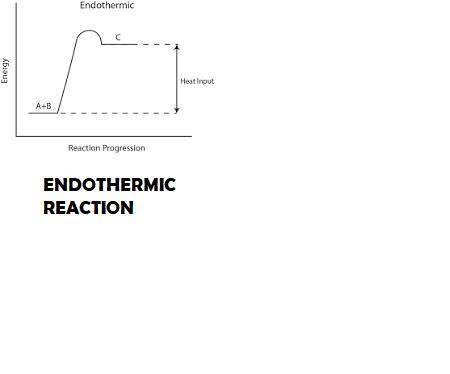
The dissolution of ammonium nitrate, nh4no3, in water is an endothermic process. since the calorimeter is not a perfect insulator, will the enthalpy of solution for ammonium nitrate be reported as too high or too low if this heat change is ignored? explain

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 09:00
How are isotopes of the same chemical element alike? how are they different?
Answers: 1

You know the right answer?
The dissolution of ammonium nitrate, nh4no3, in water is an endothermic process. since the calorimet...
Questions

History, 30.09.2019 18:00


Biology, 30.09.2019 18:00





Mathematics, 30.09.2019 18:00


English, 30.09.2019 18:00

Chemistry, 30.09.2019 18:00



Business, 30.09.2019 18:00


Social Studies, 30.09.2019 18:00




English, 30.09.2019 18:00