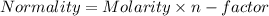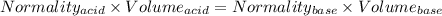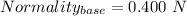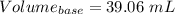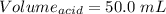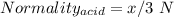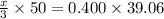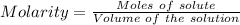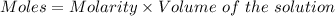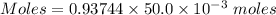Chemistry, 16.12.2019 19:31 secondcolinmills005
You are given a 1.00 g sample of an unknown tri-protic acid, which you dissolve in 50.0 ml of water containing phenolphthalein indicator. you titrate the acid solution with standardized 0.400 m koh(aq). it requires 39.06 ml of the koh solution to produce a light pink indicator color. what is the molecular weight of the unknown acid?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many moles of air are there in a human lung with a volume of 2.4 l at stp? explain your answer
Answers: 1

Chemistry, 22.06.2019 21:00
Which of these is an example of pseudoscience? a) predicting the time of sunrise based on data on position of earth b) predicting the date of the moon phases based on data on position of earth c) predicting eclipses based on the position of the sun and the moon d) predicting future events in a person's life based on the position of the moon
Answers: 1

Chemistry, 22.06.2019 23:00
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1

You know the right answer?
You are given a 1.00 g sample of an unknown tri-protic acid, which you dissolve in 50.0 ml of water...
Questions

Social Studies, 15.08.2019 09:10

History, 15.08.2019 09:10

Mathematics, 15.08.2019 09:10


English, 15.08.2019 09:10

Mathematics, 15.08.2019 09:10






Biology, 15.08.2019 09:10

History, 15.08.2019 09:10



Mathematics, 15.08.2019 10:10


English, 15.08.2019 10:10

English, 15.08.2019 10:10

English, 15.08.2019 10:10