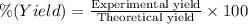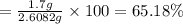Chemistry, 16.12.2019 18:31 lydiakegg454
Aspirin (c9h8o4) may be synthesized by mixing salicylic acid (c7h6o3) with excess acetic anhydride (c4h6o3).
the balanced equation is c7h6o3 + c4h6o3 c9h8o4 + c2h4o2 when 2.0 g of salicylic acid is mixed with excess acetic anhydride, 1.7 g of aspirin is collected.
what is the percent yield for this reaction?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 22.06.2019 17:20
The small bags of silica gel you often see in a new shoe box are placed there to control humidity. despite its name, silica gel is a solid. it is a chemically inert, highly porous, amorphous form of sio2. because water vapor readily adsorbs onto the surface of silica gel, it acts as a desiccant. despite not knowing mechanistic details of the adsorption of water onto silica gel, from the information provided you should be able to make an educated guess about the thermodynamic characteristics of the process. predict the signs for δg, δh, and δs for the adsorption of water.
Answers: 2
You know the right answer?
Aspirin (c9h8o4) may be synthesized by mixing salicylic acid (c7h6o3) with excess acetic anhydride (...
Questions


Physics, 27.10.2020 02:30

Mathematics, 27.10.2020 02:30



Business, 27.10.2020 02:30

English, 27.10.2020 02:30

Business, 27.10.2020 02:30


Mathematics, 27.10.2020 02:30

Social Studies, 27.10.2020 02:30


Mathematics, 27.10.2020 02:30

Mathematics, 27.10.2020 02:30



Social Studies, 27.10.2020 02:30

Biology, 27.10.2020 02:30

Mathematics, 27.10.2020 02:30

Engineering, 27.10.2020 02:30

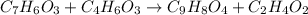
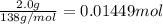
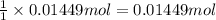 of asprin
of asprin