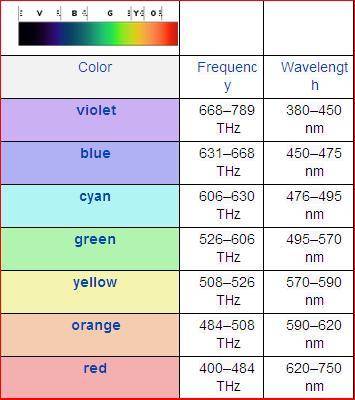
Chemistry, 14.12.2019 08:31 Nathaliasmiles
Astudent analyzes the emission spectrum of an unknown element and observes strong lines at the following wavelengths:
656 nm, 486 nm, 434 nm.
which absorption spectrum is the student most likely viewing?
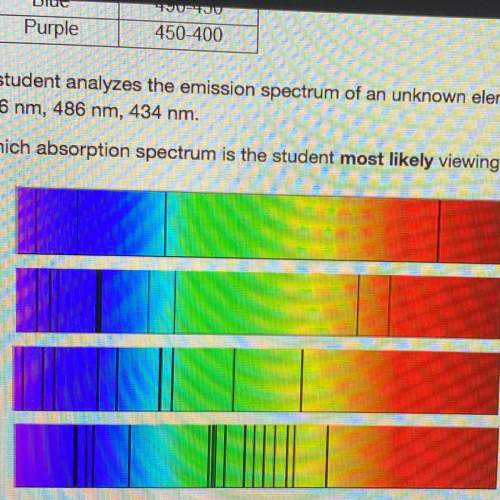

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:20
1. suppose a reaction mixture, when diluted with water, afforded 300 ml of an aqueous solution of 30 g of the reaction product malononitrile [ch2(cn)2], which is to be isolated by extraction with ether. the solubility of malononitrile in ether at room temperature is 20.0 g/100 ml, and in water is 13.3 g/100 ml. what weight of malononitrile would be recovered by extraction with (a) three 100-ml portions of ether and (b) one 300-ml portion of ether? suggestion: for each extraction, let x equal the weight extracted into the ether layer. in part (a), the concentration in the ether layer is x/100 and in the water layer is (30 x)/300; the ratio of these quantities is equal to k 20/13.3.
Answers: 2

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 21:30
Under which circumstances are kp and kc equal for the reaction aa(g)+bb(g)⇌cc(g)+dd(g)?
Answers: 2

Chemistry, 23.06.2019 03:30
Astudent uses universal ph paper to find the ph of three solutions . solution a has a ph of 5 solution b has a ph of 11 and solution c has a ph of 7 identify which solution is acidic which solution is neutral and which solution is basic
Answers: 1
You know the right answer?
Astudent analyzes the emission spectrum of an unknown element and observes strong lines at the follo...
Questions


History, 17.12.2020 18:10





Biology, 17.12.2020 18:10




Mathematics, 17.12.2020 18:10

Chemistry, 17.12.2020 18:10





Chemistry, 17.12.2020 18:10

Mathematics, 17.12.2020 18:10


Mathematics, 17.12.2020 18:10