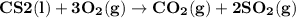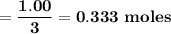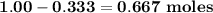Carbon disulfide burns in oxygen to yield carbon dioxide and sulfur dioxide according to the following chemical equation. cs2(l) + 3o2(g) → co2(g) + 2so2(g)
a. if 1.00 mol cs2 reacts with 1.00 mol o2, identify the limiting reactant.
b. how many moles of excess reactant remain?
c. how many moles of each product are formed?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 22.06.2019 06:30
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3

Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
Carbon disulfide burns in oxygen to yield carbon dioxide and sulfur dioxide according to the followi...
Questions





Chemistry, 17.07.2019 16:30



History, 17.07.2019 16:30

Mathematics, 17.07.2019 16:30



Biology, 17.07.2019 16:30

Social Studies, 17.07.2019 16:30

Mathematics, 17.07.2019 16:30

Mathematics, 17.07.2019 16:30

Biology, 17.07.2019 16:30



History, 17.07.2019 16:30