
Chemistry, 13.12.2019 01:31 karinagramirezp072gb
Consider the following chemical equation. how many moles of seo3 can be produced from 35
g of oxygen and excess selenium?
25e +302 - 25e03

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 23.06.2019 02:30
Asubstance is held in an open container. its particles move past one another at random speeds but do not leave the container. heat is removed from the system, and the particles slow down. when enough heat is removed, the particles no longer have enough speed to overcome the weak attractive forces between them. when this happens, the substance enters its solid state. the process described above is known as .
Answers: 3
You know the right answer?
Consider the following chemical equation. how many moles of seo3 can be produced from 35
g of...
g of...
Questions

History, 21.05.2020 02:06



Mathematics, 21.05.2020 02:06

Mathematics, 21.05.2020 02:06

Social Studies, 21.05.2020 02:06


Spanish, 21.05.2020 02:07


Mathematics, 21.05.2020 02:07


Mathematics, 21.05.2020 02:07

Mathematics, 21.05.2020 02:07


Mathematics, 21.05.2020 02:07

History, 21.05.2020 02:07



Biology, 21.05.2020 02:07

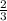 moles of SeO3
moles of SeO3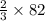 g of SeO3
g of SeO3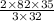 g of SeO3
g of SeO3 

