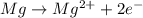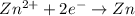Chemistry, 27.01.2020 05:31 joybeth9591
What gets oxidized in a galvanic cell made with magnesium and zinc electrodes?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:30
If anyone would be able to me out with these three questions it would be these are from the chem 2202 course.
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
What gets oxidized in a galvanic cell made with magnesium and zinc electrodes?...
Questions

English, 19.11.2020 01:00

Mathematics, 19.11.2020 01:00

Law, 19.11.2020 01:00

SAT, 19.11.2020 01:00


Mathematics, 19.11.2020 01:00

Chemistry, 19.11.2020 01:00

Biology, 19.11.2020 01:00

English, 19.11.2020 01:00

Computers and Technology, 19.11.2020 01:00



Mathematics, 19.11.2020 01:00

History, 19.11.2020 01:00

English, 19.11.2020 01:00

History, 19.11.2020 01:00




Mathematics, 19.11.2020 01:00

![E^0_{[Mg^{2+}/Mg]}= -2.37V](/tpl/images/0472/1081/24fc1.png)
![E^0_{[Zn^{2+}/Zn]}=-0.76V](/tpl/images/0472/1081/4cd18.png)