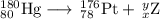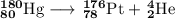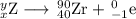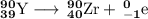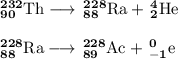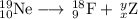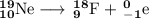Chemistry, 12.12.2019 02:31 brooke012002
Write a balanced equation for each of the following nuclear reactions: (a) mercury-180 decays into platinum-176 (b) zirconium-90 and an electron are produced by the decay of an unstable nucleus (c) thorium-232 decays and produces an alpha particle and a radium-228 nucleus, which decays into actinium-228 by beta decay (d) neon-19 decays into fluorine-19

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 17:30
What type of organic molecule comprises the majority of a potato?
Answers: 1

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3
You know the right answer?
Write a balanced equation for each of the following nuclear reactions: (a) mercury-180 decays into...
Questions

Mathematics, 17.06.2021 16:50


Mathematics, 17.06.2021 16:50






World Languages, 17.06.2021 16:50


History, 17.06.2021 16:50

English, 17.06.2021 16:50

English, 17.06.2021 16:50

Mathematics, 17.06.2021 16:50


Mathematics, 17.06.2021 16:50

Mathematics, 17.06.2021 16:50

English, 17.06.2021 16:50


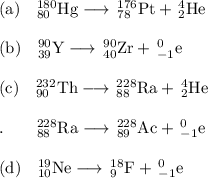
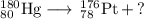
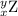 , where x = the atomic number, y = the mass number, and Z = the symbol of the element .
, where x = the atomic number, y = the mass number, and Z = the symbol of the element .