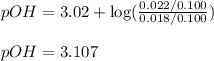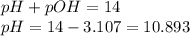Chemistry, 09.12.2019 21:31 emilyturchon
Calculate the ph of a solution prepared by adding 20.0 ml of 0.100 m hcl to 80.0 ml of a buffer that is comprised of 0.25 m c2h5nh2 and 0.25 m c2h5nh3cl. kb of c2h5nh2 = 9.5 x 10-4.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:50
Which of these reactions are redox reactions? check all that apply.cd + hcl → cdcl2 + h2cucl2 + na2s → 2nacl + cuscaco3 → cao + co2 2zns + 3o2 → 2zno + 2so2 ch4 + 2o2 → co2 + 2h2o
Answers: 3

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
Calculate the ph of a solution prepared by adding 20.0 ml of 0.100 m hcl to 80.0 ml of a buffer that...
Questions


English, 28.09.2019 06:50

Mathematics, 28.09.2019 06:50



History, 28.09.2019 06:50



Mathematics, 28.09.2019 06:50








English, 28.09.2019 06:50



Mathematics, 28.09.2019 06:50

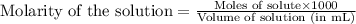
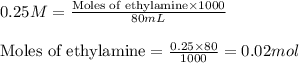
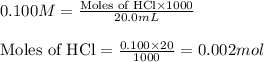
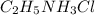 :
: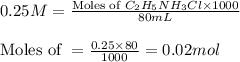
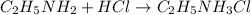
![pOH=pK_b+\log(\frac{[salt]}{[base]})](/tpl/images/0410/5365/db86b.png)
![pOH=pK_b+\log(\frac{[C_2H_5NH_3Cl]}{[C_2H_5NH_2]})](/tpl/images/0410/5365/2dd1c.png)
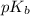 = negative logarithm of base dissociation constant of ethylamine =
= negative logarithm of base dissociation constant of ethylamine = 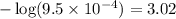
![[C_2H_5NH_3Cl]=\frac{0.022}{0.100}](/tpl/images/0410/5365/d3f48.png)
![[C_2H_5NH_2]=\frac{0.018}{0.100}](/tpl/images/0410/5365/5f984.png)