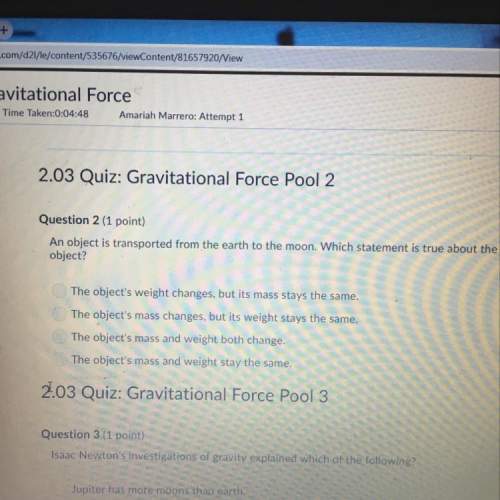
An acetic acid buffer solution is required to have a ph of 5.27. you have a solution that contains 0.01 mol of acetic acid. what molarity of sodium acetate will you need to add to the solution? the pka of acetic acid is 4.74. show all calculations in your answer.
in this experiment, you will make an acetic acid buffer by adding sodium hydroxide (naoh) to commercial vinegar. commercial vinegar is usually comprised of 5% acetic acid by volume. the sodium hydroxide will react with the acetic acid to form sodium acetate (ch₃coona). see equation below.
ch₃cooh(aq) + naoh(aq) > ch₃coona(aq) + h2o(l)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3


Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
An acetic acid buffer solution is required to have a ph of 5.27. you have a solution that contains 0...
Questions

Biology, 05.07.2019 02:00


Geography, 05.07.2019 02:00


Mathematics, 05.07.2019 02:00



English, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00


Mathematics, 05.07.2019 02:00




Social Studies, 05.07.2019 02:00



History, 05.07.2019 02:00

Mathematics, 05.07.2019 02:00

Computers and Technology, 05.07.2019 02:00