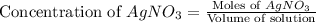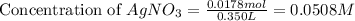Chemistry, 06.12.2019 19:31 orlando19882000
Suppose a 2.95 g of potassium iodide is dissolved in 350. ml of a 62.0 m m aqueous solution of silver nitrate. calculate the final molarity of iodide anion in the solution. you can assume the volume of the solution doesn't change when the potassium iodide is dissolved in it. be sure your answer has the correct number of significant digits

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2

Chemistry, 23.06.2019 09:00
Need ! assume that the variables x and y are directly related. if k = 8, what is the value for each of the following points? be sure and record your data to be used in the following problem. x y k 0.
Answers: 2

Chemistry, 23.06.2019 10:40
Question 17 hydrogen is manufactured on an industrial scale by this sequence of reactions: +ch4gh2og ⇌ +cog3h2g k1 +cogh2og ⇌ +co2gh2g k2 the net reaction is: +ch4g2h2og ⇌ +co2g4h2g k write an equation that gives the overall equilibrium constant k in terms of the equilibrium constants k1 and k2. if you need to include any physical constants, be sure you use their standard symbols, which you'll find in the aleks calculator.
Answers: 2
You know the right answer?
Suppose a 2.95 g of potassium iodide is dissolved in 350. ml of a 62.0 m m aqueous solution of silve...
Questions



Social Studies, 27.08.2020 18:01

English, 27.08.2020 18:01



History, 27.08.2020 18:01

Chemistry, 27.08.2020 19:01


English, 27.08.2020 19:01



Mathematics, 27.08.2020 19:01



Mathematics, 27.08.2020 19:01

English, 27.08.2020 19:01

Biology, 27.08.2020 19:01


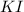 and
and 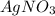 .
.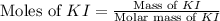
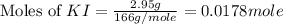
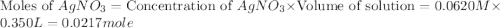
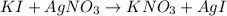
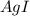
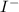 anion = Moles of
anion = Moles of 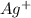 cation = 0.0178 moles
cation = 0.0178 moles