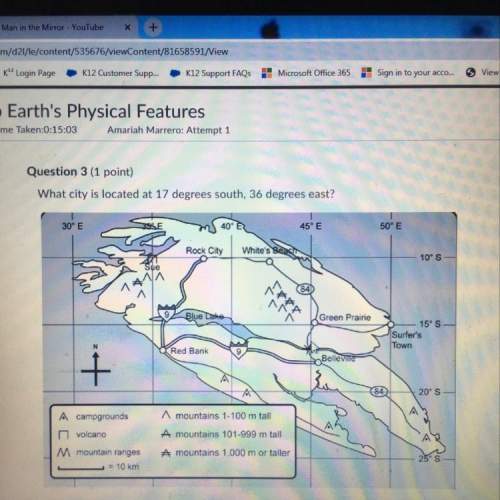
Calculate the lattice energy for naf (s) given the following
sublimation of energy +109...

Chemistry, 06.12.2019 19:31 DiamondW8526
Calculate the lattice energy for naf (s) given the following
sublimation of energy +109 kj/mol
hf for f (g) + 77 kj/mol
first ionization energy of na (g) +495
electron affinity of f(g) -328
enthalpy of formation of naf(s) -570 kj/mol
a. -177
b. 192
c. -804
d. -1047
e. -923

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1
You know the right answer?
Questions

Social Studies, 07.04.2020 03:19

Mathematics, 07.04.2020 03:19

Spanish, 07.04.2020 03:19

Mathematics, 07.04.2020 03:19





Mathematics, 07.04.2020 03:20

Health, 07.04.2020 03:20


Social Studies, 07.04.2020 03:20


History, 07.04.2020 03:20

History, 07.04.2020 03:20



Mathematics, 07.04.2020 03:20

Mathematics, 07.04.2020 03:20

Mathematics, 07.04.2020 03:20