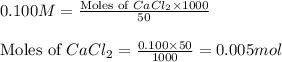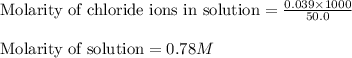Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 23:30
Aweight lifter raises a 1600 n barbell to a height of 2.0 meters. how much work was done? w = fd a) 30 joules b) 3000 joules c) 320 joules d) 3200 joules
Answers: 2

Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1
You know the right answer?
Asolution is prepared by adding 1.70 g of solid nacl to 50.0 ml of 0.100 m cacl2. what is the molari...
Questions


Social Studies, 18.09.2019 14:30

Mathematics, 18.09.2019 14:30

Computers and Technology, 18.09.2019 14:30

Mathematics, 18.09.2019 14:30

English, 18.09.2019 14:30

Chemistry, 18.09.2019 14:30


History, 18.09.2019 14:30

History, 18.09.2019 14:30

Mathematics, 18.09.2019 14:30


History, 18.09.2019 14:30

Geography, 18.09.2019 14:30





Mathematics, 18.09.2019 14:30

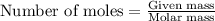
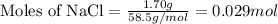
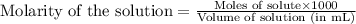 .....(1)
.....(1)