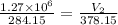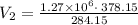Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

You know the right answer?
Ahot air balloon filled with 1.27x10^6l of an ideal gas on a cool morning of 11 degrees c. the air i...
Questions

Mathematics, 16.07.2019 05:00


History, 16.07.2019 05:00





Mathematics, 16.07.2019 05:00

Computers and Technology, 16.07.2019 05:00


Chemistry, 16.07.2019 05:00




Computers and Technology, 16.07.2019 05:00



Computers and Technology, 16.07.2019 05:00


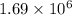 L
L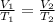
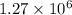 L
L