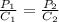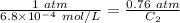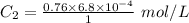Chemistry, 04.12.2019 00:31 fireman59937
The solubility of nitrogen gas at 25 ◦c and 1 atm is 6.8×10−4 mol/l. if the partial pressure of nitrogen gas in air is 0.76 atm, what is the concentration (molarity) of dissolved nitrogen?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The best solution for preventing harm to people and pets from severe hurricanes involves determining and warning residents about what
Answers: 1

Chemistry, 22.06.2019 20:30
Consider the following unbalanced equation for the combustion of hexane: αc6h14(g)+βo2(g)→γco2(g)+δh2o(g) part a balance the equation. give your answer as an ordered set of numbers α, β, γ, use the least possible integers for the coefficients. α α , β, γ, δ = nothing request answer part b determine how many moles of o2 are required to react completely with 5.6 moles c6h14. express your answer using two significant figures. n n = nothing mol request answer provide feedback
Answers: 2

Chemistry, 23.06.2019 03:30
Calculate the ph of a .10m nh4cl solution. the kb value for nh3 is 1.8×10^-5
Answers: 1

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1
You know the right answer?
The solubility of nitrogen gas at 25 ◦c and 1 atm is 6.8×10−4 mol/l. if the partial pressure of nitr...
Questions



Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

History, 07.10.2020 06:01



English, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01



Mathematics, 07.10.2020 06:01




Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01