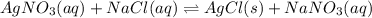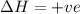Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2


Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3

Chemistry, 23.06.2019 06:30
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1
You know the right answer?
For the endothermic reaction below, using lechatlier's principle, predict which way the equilibrium...
Questions

Computers and Technology, 02.11.2020 17:00

English, 02.11.2020 17:00





Computers and Technology, 02.11.2020 17:00

Mathematics, 02.11.2020 17:00

Mathematics, 02.11.2020 17:00



Mathematics, 02.11.2020 17:00

Computers and Technology, 02.11.2020 17:00




Business, 02.11.2020 17:00


History, 02.11.2020 17:00

Mathematics, 02.11.2020 17:00