
Chemistry, 29.11.2019 02:31 mathman783
Given that δ h ∘ f [ br ( g ) ] = 111.9 kj ⋅ mol − 1 δ h ∘ f [ c ( g ) ] = 716.7 kj ⋅ mol − 1 δ h ∘ f [ cbr 4 ( g ) ] = 29.4 kj ⋅ mol − 1 calculate the average molar bond enthalpy of the carbon‑bromine bond in a cbr 4 molecule.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1

You know the right answer?
Given that δ h ∘ f [ br ( g ) ] = 111.9 kj ⋅ mol − 1 δ h ∘ f [ c ( g ) ] = 716.7 kj ⋅ mol − 1 δ h ∘...
Questions

English, 22.09.2019 06:00




Mathematics, 22.09.2019 06:00

Biology, 22.09.2019 06:00




Social Studies, 22.09.2019 06:00

Mathematics, 22.09.2019 06:00

Mathematics, 22.09.2019 06:10

Physics, 22.09.2019 06:10

History, 22.09.2019 06:10


Mathematics, 22.09.2019 06:10


Biology, 22.09.2019 06:10

Chemistry, 22.09.2019 06:10

History, 22.09.2019 06:10

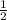 Br2(g) ⇒ Br(g) , Δ H ∘ = 111.9 kJ ⋅ mol − 1
Br2(g) ⇒ Br(g) , Δ H ∘ = 111.9 kJ ⋅ mol − 1 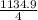 = 283.725 kJ ⋅ mol − 1
= 283.725 kJ ⋅ mol − 1

