
Chemistry, 28.11.2019 04:31 dbn4everloved8
A1.00 g sample of n-hexane (c6h14) undergoes complete combustion with excess o2 in a bomb calorimeter. the temperature of the 1502 g of water surrounding the bomb rises from 22.64°c to 29.30°c. the heat capacity of the hardware component of the calorimeter (everything that is not water) is 4042 j/°c. what is δu for the combustion of n-c6h14? one mole of n-c6h14 is 86.1 g. the specific heat of water is 4.184 j/g·°c.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:20
If i can still dissolve more sugar into the solution at a certain temperature what would i call that solution
Answers: 3

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
A1.00 g sample of n-hexane (c6h14) undergoes complete combustion with excess o2 in a bomb calorimete...
Questions

Mathematics, 07.01.2021 21:30

Mathematics, 07.01.2021 21:30


Mathematics, 07.01.2021 21:30

Mathematics, 07.01.2021 21:30


Mathematics, 07.01.2021 21:30


History, 07.01.2021 21:30

Social Studies, 07.01.2021 21:30

Mathematics, 07.01.2021 21:40

Engineering, 07.01.2021 21:40

Mathematics, 07.01.2021 21:40

Mathematics, 07.01.2021 21:40

Mathematics, 07.01.2021 21:40

Mathematics, 07.01.2021 21:40


Social Studies, 07.01.2021 21:40


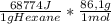 = -5,921x10⁶J/mol
= -5,921x10⁶J/mol

