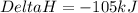Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:50
How does a scientist the government? a. the scientist tells people in society what to do. b. the scientist determines the policies that the government spends money on. c. the scientist provides unbiased information to the government. d. the scientist makes laws based on his or her research results.
Answers: 1

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3
You know the right answer?
Nitrogen and hydrogen combine to form ammonia in the haber process. calculate (in kj) the standard e...
Questions

History, 01.12.2021 01:30



Computers and Technology, 01.12.2021 01:30




Engineering, 01.12.2021 01:30

Computers and Technology, 01.12.2021 01:30


Chemistry, 01.12.2021 01:30




SAT, 01.12.2021 01:30


Geography, 01.12.2021 01:30

Business, 01.12.2021 01:30


History, 01.12.2021 01:30

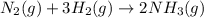
![\Delta H=\sum [n\times B.E(reactant)]-\sum [n\times B.E(product)]](/tpl/images/0392/9421/42942.png)
![\Delta H=[(n_{N_2}\times B.E_{N_2})+(n_{H_2}\times B.E_{H_2}) ]-[(n_{NH_3}\times B.E_{NH_3})]](/tpl/images/0392/9421/acc53.png)
![\Delta H=[(n_{N_2}\times B.E_{N\equiv N})+(n_{H_2}\times B.E_{H-H}) ]-[(n_{NH_3}\times 3\times B.E_{N-H})]](/tpl/images/0392/9421/03d6a.png)
![\Delta H=[(1\times 945)+(3\times 432)]-[(2\times 3\times 391)]](/tpl/images/0392/9421/a2882.png)