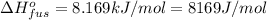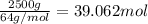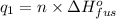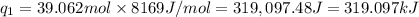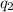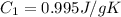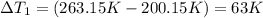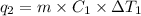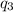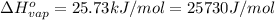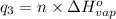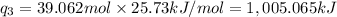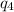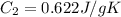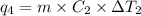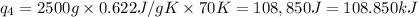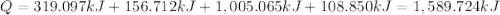Sulfur dioxide is produced in enormous amounts for sulfuric acid production. it melts at - 73˚c and boils at - 10˚c. its δh˚fus is 8.619 kj/mol and its δh˚vap is 25.73 kj/mol. the specific heat capacities of the liquid and the gas are 0.995 j/g·k and 0.622 j/g·k, respectively. how much heat is required to convert 2.500 kg of solid so₂ at the melting point to a gas at 60˚c?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 04:00
How do scientists think that gravity affected the formation of our solar system?
Answers: 1

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2
You know the right answer?
Sulfur dioxide is produced in enormous amounts for sulfuric acid production. it melts at - 73˚c and...
Questions

SAT, 26.01.2022 07:40


Chemistry, 26.01.2022 07:40

Computers and Technology, 26.01.2022 07:40

Mathematics, 26.01.2022 07:40


Mathematics, 26.01.2022 07:40








Mathematics, 26.01.2022 07:40

Mathematics, 26.01.2022 07:40