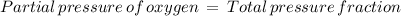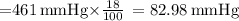Chemistry, 22.11.2019 09:31 nevaehkirk1997
The atmospheric pressure at this altitude is 461 mmhg. assuming that the atmosphere is 18% oxygen (by volume), calculate the partial pressure of o2.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1


Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
The atmospheric pressure at this altitude is 461 mmhg. assuming that the atmosphere is 18% oxygen (b...
Questions



English, 04.03.2021 02:20


Mathematics, 04.03.2021 02:20


Mathematics, 04.03.2021 02:20



Mathematics, 04.03.2021 02:20




History, 04.03.2021 02:20

Chemistry, 04.03.2021 02:20




Mathematics, 04.03.2021 02:20