Chemistry, 21.11.2019 22:31 Redhead667
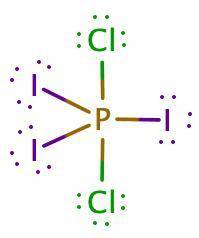
Pi3cl2 is a nonpolar molecule. based on this information, determine the i−p−i bond angle, the cl−p−cl bond angle, and the i−p−cl bond angle. enter the number of degrees of the i−p−i, cl−p−cl, and i−p−cl bond angles, separated by commas (e. g., 30,45,90)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2
You know the right answer?
Pi3cl2 is a nonpolar molecule. based on this information, determine the i−p−i bond angle, the cl−p−c...
Questions


Biology, 31.07.2019 09:30

Computers and Technology, 31.07.2019 09:30



Health, 31.07.2019 09:30

Geography, 31.07.2019 09:30

Mathematics, 31.07.2019 09:30



Geography, 31.07.2019 09:30

Spanish, 31.07.2019 09:30

Mathematics, 31.07.2019 09:30


English, 31.07.2019 09:30


Mathematics, 31.07.2019 09:30