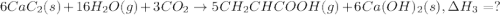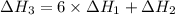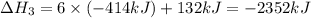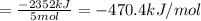Chemistry, 19.11.2019 23:31 sipstick9411
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: (s) (g) (g) (s) in the second step, acetylene, carbon dioxide and water react to form acrylic acid: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest .

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of...
Questions




Mathematics, 25.02.2021 20:00

English, 25.02.2021 20:00



Chemistry, 25.02.2021 20:00


Mathematics, 25.02.2021 20:00

Mathematics, 25.02.2021 20:00

Mathematics, 25.02.2021 20:00


Mathematics, 25.02.2021 20:00

Mathematics, 25.02.2021 20:00

Mathematics, 25.02.2021 20:00

Chemistry, 25.02.2021 20:00


Mathematics, 25.02.2021 20:00

Mathematics, 25.02.2021 20:00

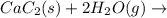
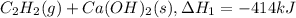 ..[1]
..[1]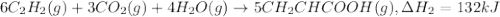 ..[2]
..[2]