In this illustration, which group highlights, or which groups highlight, nonmetals?
...

Chemistry, 18.11.2019 21:31 SKYBLUE1015
In this illustration, which group highlights, or which groups highlight, nonmetals?
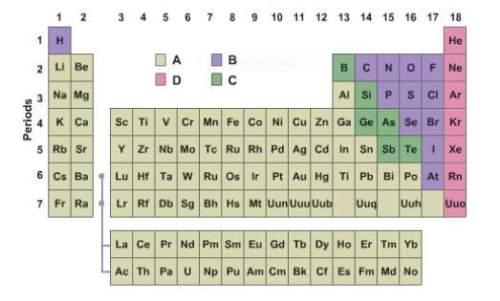

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3
You know the right answer?
Questions

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Business, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Biology, 21.10.2019 17:30

Physics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30

Mathematics, 21.10.2019 17:30


Mathematics, 21.10.2019 17:30

World Languages, 21.10.2019 17:30

Business, 21.10.2019 17:30

Chemistry, 21.10.2019 17:30



